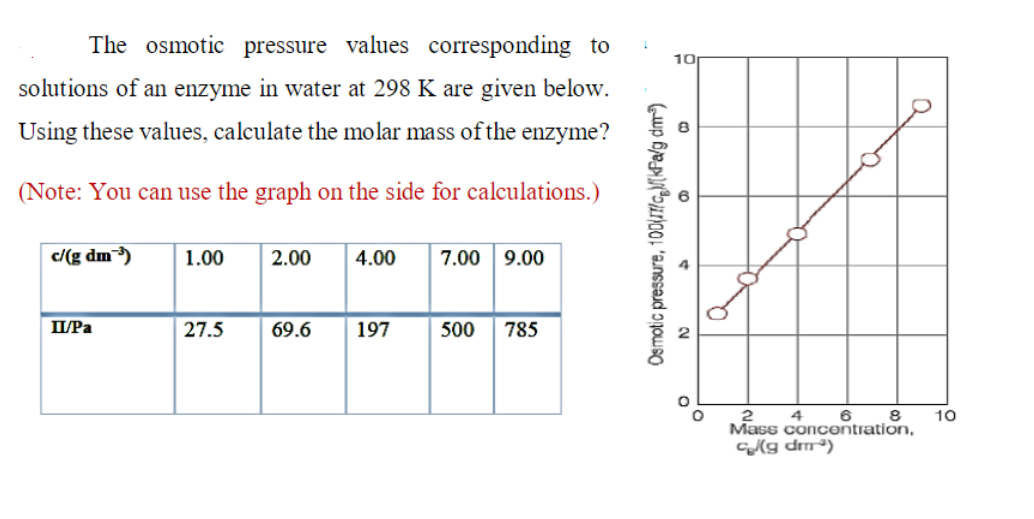
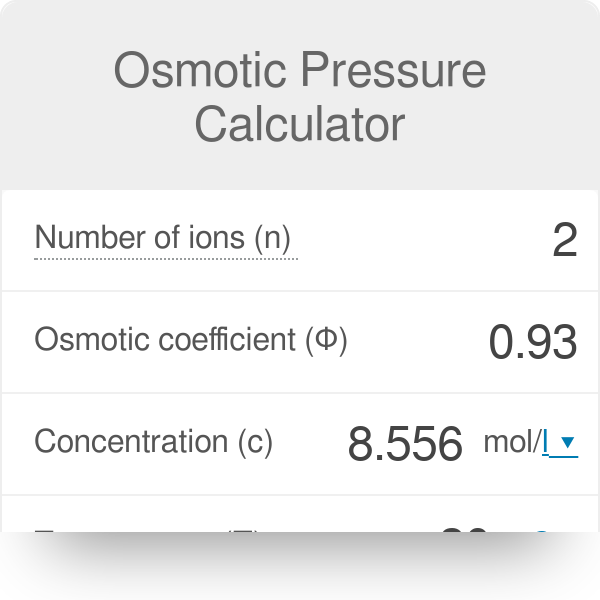
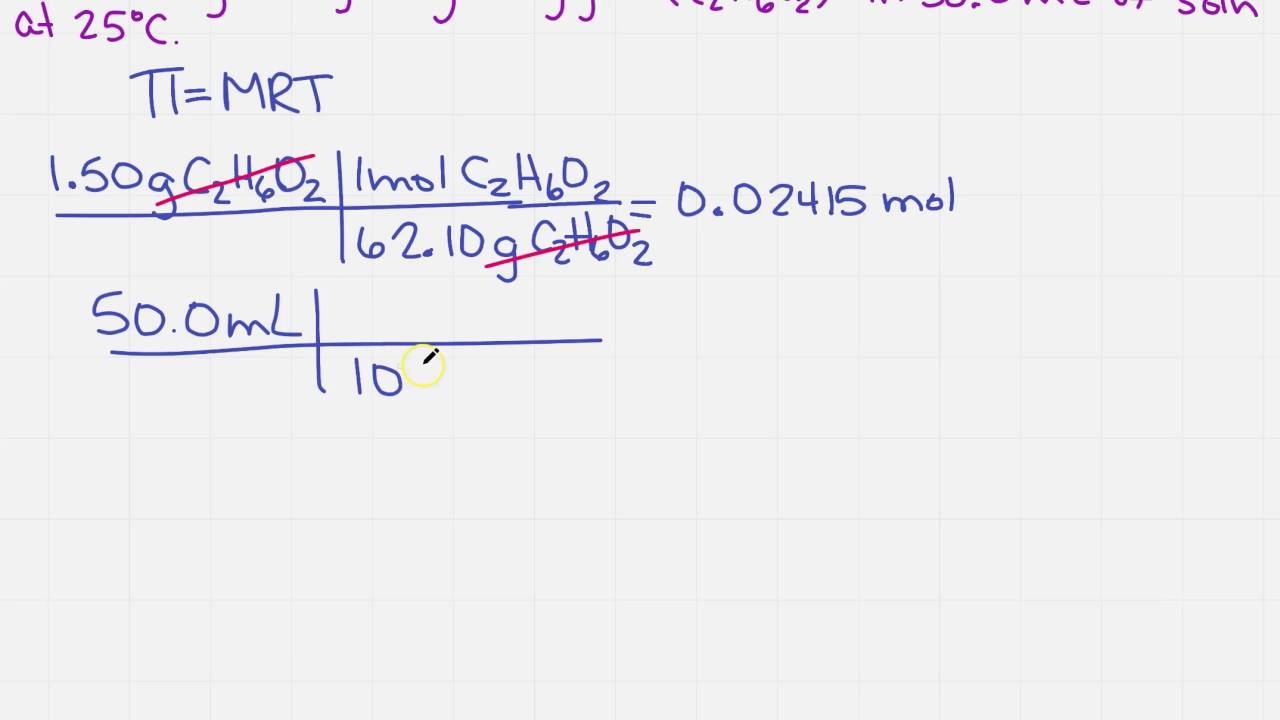Osmotic Pressure Formula & Examples | What is Osmotic Pressure? - Video & Lesson Transcript | Study.com
![For a 5% solution of urea (Molar mass - 60 g/mol), calculate the osmotic pressure at 300 K. [R = 0.0821 L atm K^(-1) mol^(-1)] For a 5% solution of urea (Molar mass - 60 g/mol), calculate the osmotic pressure at 300 K. [R = 0.0821 L atm K^(-1) mol^(-1)]](https://d10lpgp6xz60nq.cloudfront.net/ss/web/1522124.jpg)
For a 5% solution of urea (Molar mass - 60 g/mol), calculate the osmotic pressure at 300 K. [R = 0.0821 L atm K^(-1) mol^(-1)]

Calculate the osmotic pressure of 4.5g of glucose (Molar mass = 180 ) dissolved in 100ml of water at 298K .(Given : R = 0.0821Latmmol^-1k^-1

Hi need help! in calculating the osmotic pressure of an RO system using the formula, does the solute conc. mean in the feed or permeate? : r/WaterTreatment
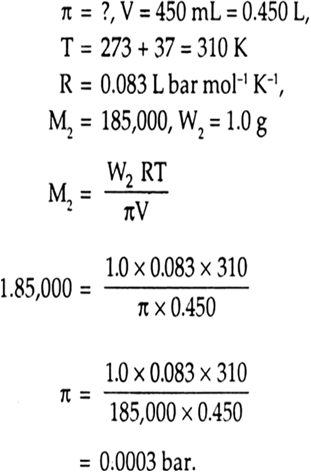
Calculate the osmotic pressure in pascals exerted by a solution prepared by dissolving 1.0 g of polymer of molar mass 185,000 in 450 mL at 370C. from Chemistry Solutions Class 12 CBSE

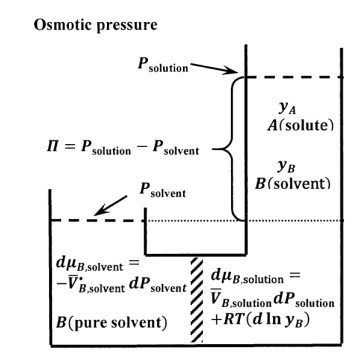
13.7 Osmotic Pressure –Osmosis, osmotic pressure calculations including molar mass determination, colligative properties, practical applications (red blood. - ppt download

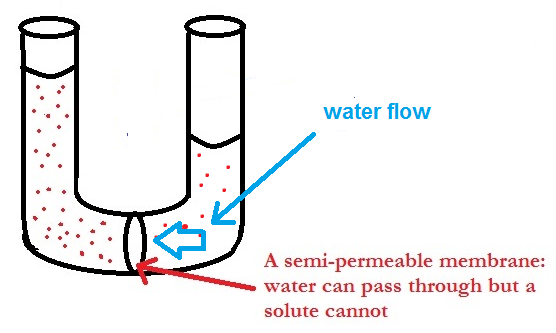
Twitter 上的DocMD #LENI2022 ✊🏼🇵🇭 💗:"25.Osmotic Pressure-pressure needed to prevent osmosis, which is the spontaneous net movement of solvent molecules through a semi-permeable membrane in to a region of higher solute concentrations.