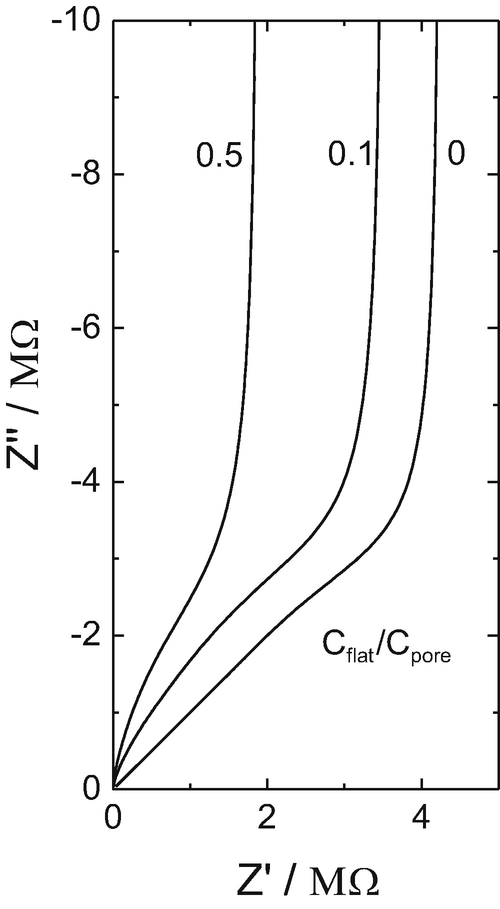
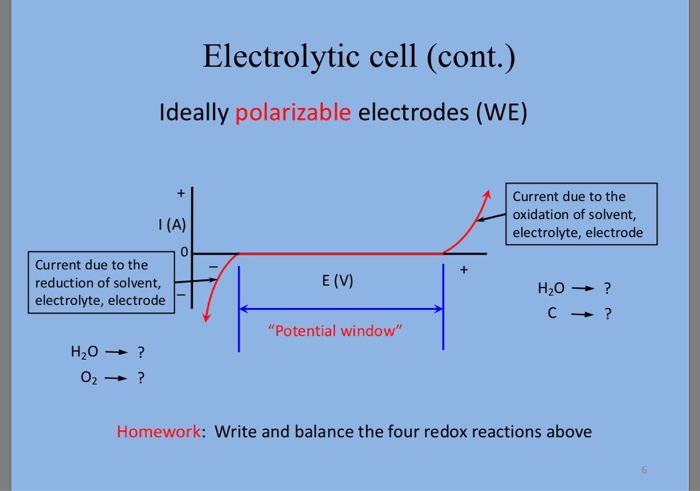
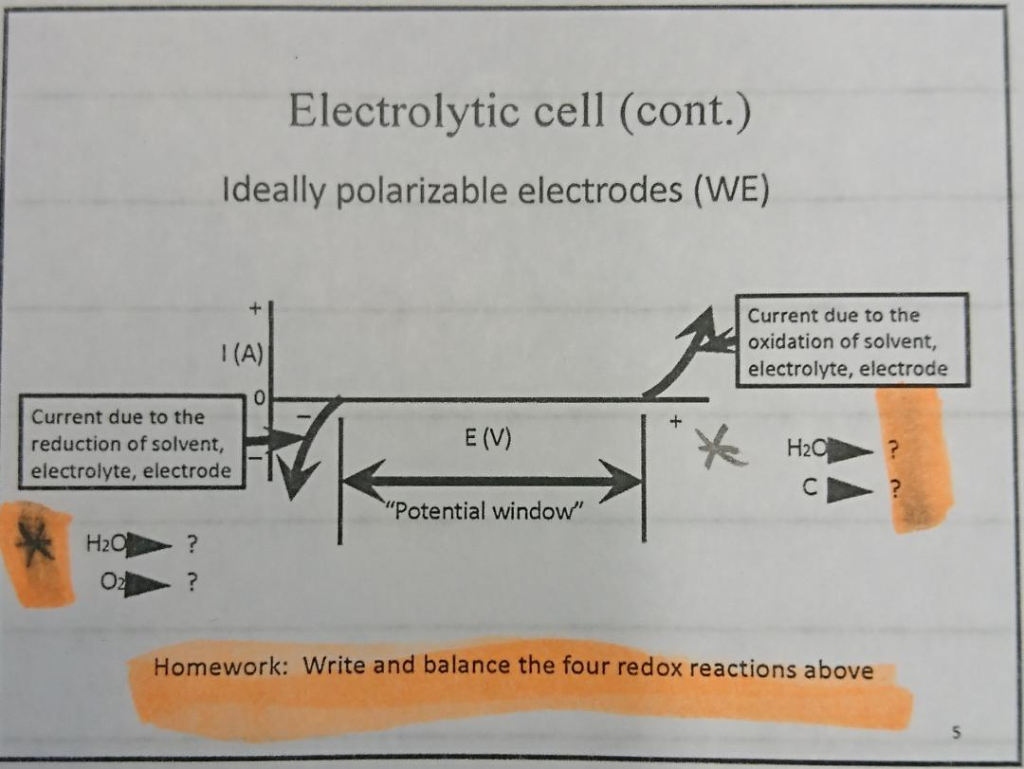
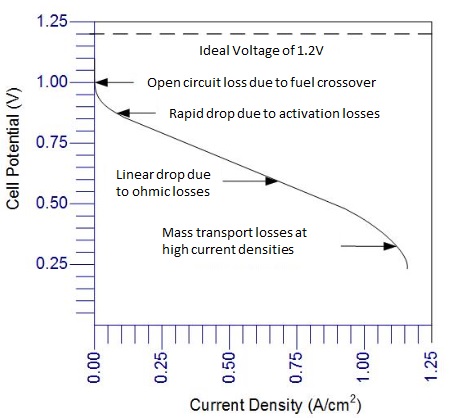
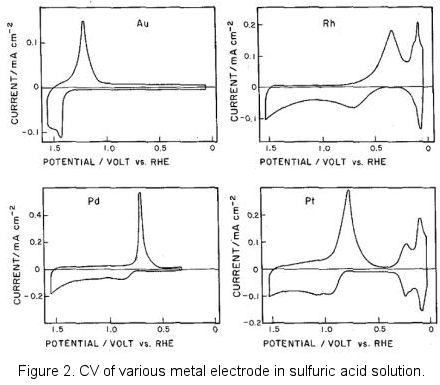
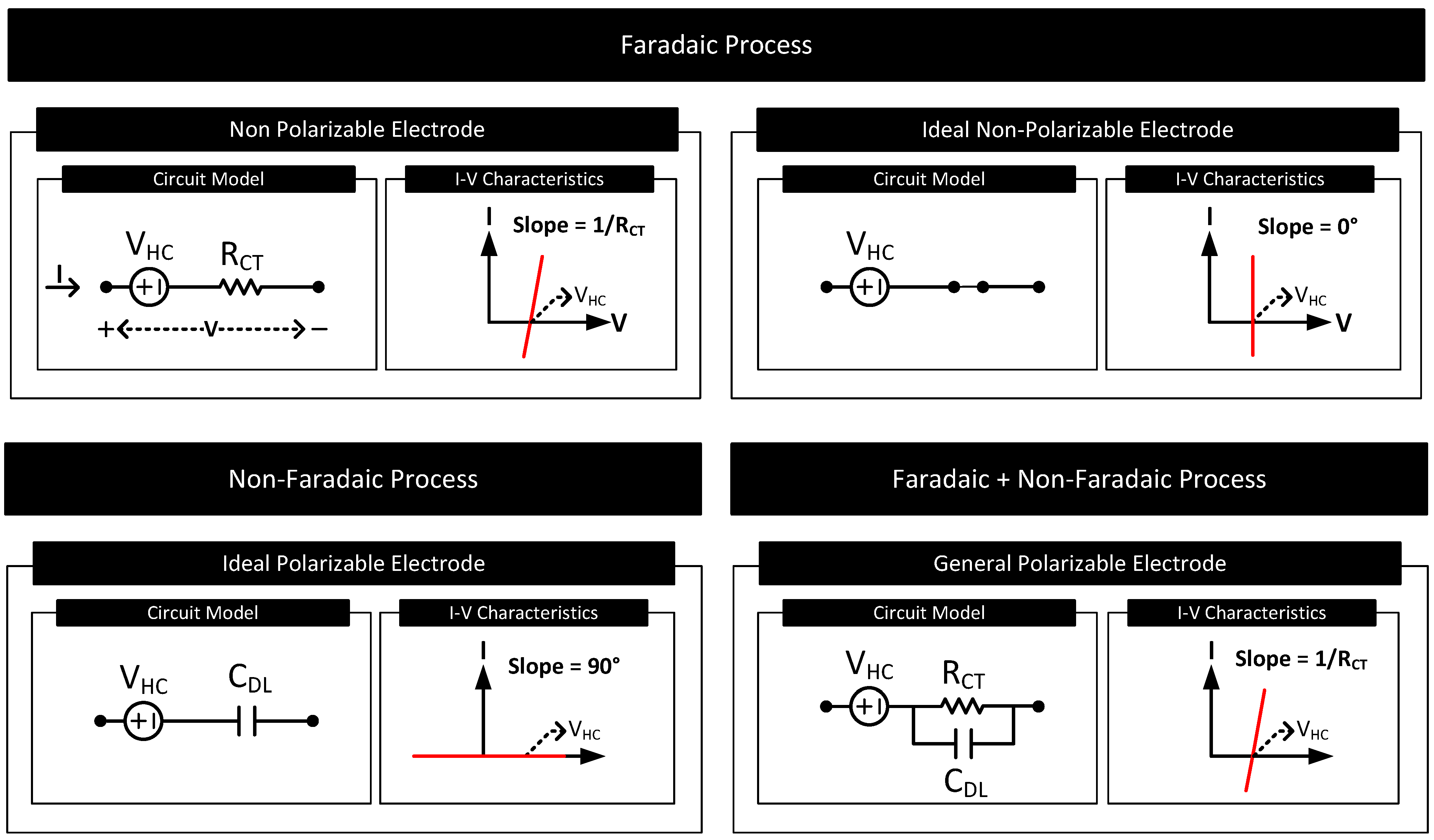
CHM 432: ADVANCED ELECTROCHEMISTRY CONTENTS 1.0. ELECTRICAL DOUBLE LAYER • Faradaic and Non faradaic current • Mass Transfe

Study of the electrolyte-insulator-semiconductor field-effect transistor (EISFET) with applications in biosensor design - ScienceDirect

Understanding the Electric Double-Layer Structure, Capacitance, and Charging Dynamics | Chemical Reviews

SOLVED: 2 A) The mercury electrode is described as an ideally polarizable electrode Provide an explanation of an ideally polarisable electrode. What are the conditions that cause a mercury electrode to deviate

SOLVED: 47 00/0 Question 9/18 (1 p ) Answer is mandatory In an electrochemical cell, if the potential of one of the electrodes was changing even when the current is kept constant (
Nanomaterials | Free Full-Text | Human Body–Electrode Interfaces for Wide-Frequency Sensing and Communication: A Review

Difference Between Polarizable and Non Polarizable Electrode | Compare the Difference Between Similar Terms

A general thermodynamic theory of the ideal polarized electrode - Transactions of the Faraday Society (RSC Publishing)

The role of reversible and polarizable surface charge in the electro-sorption of NaCl electrolyte onto activated carbon-graphite electrode - ScienceDirect