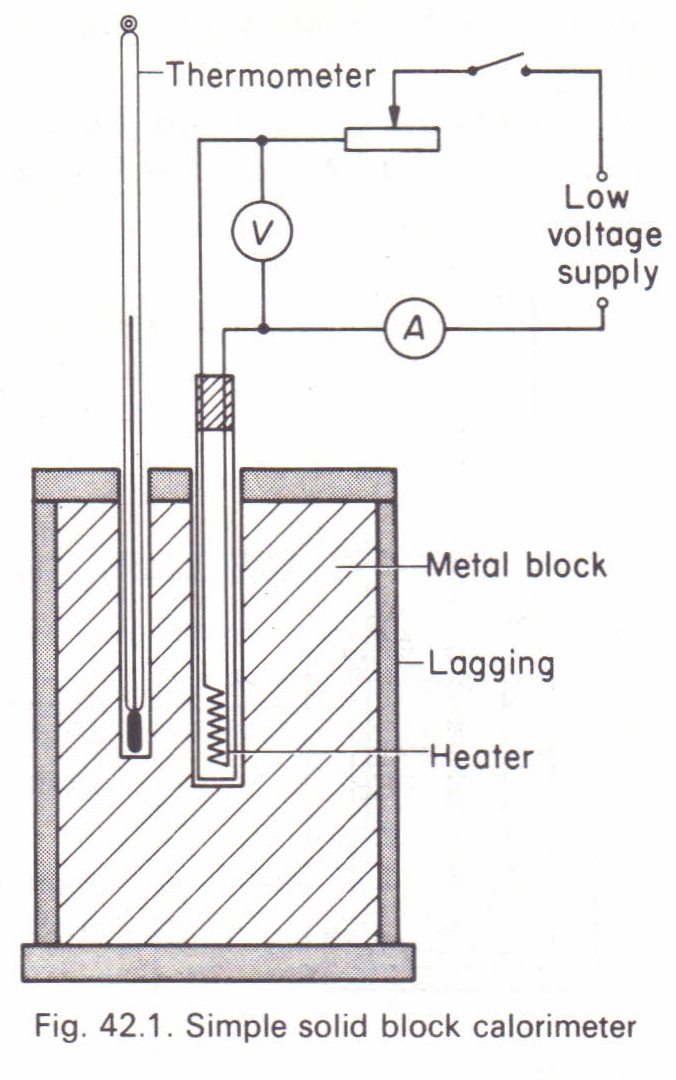
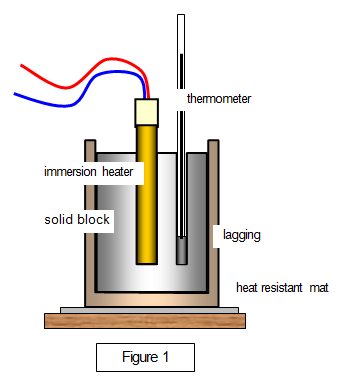
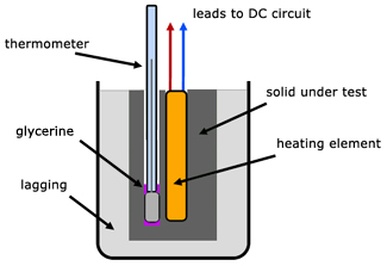
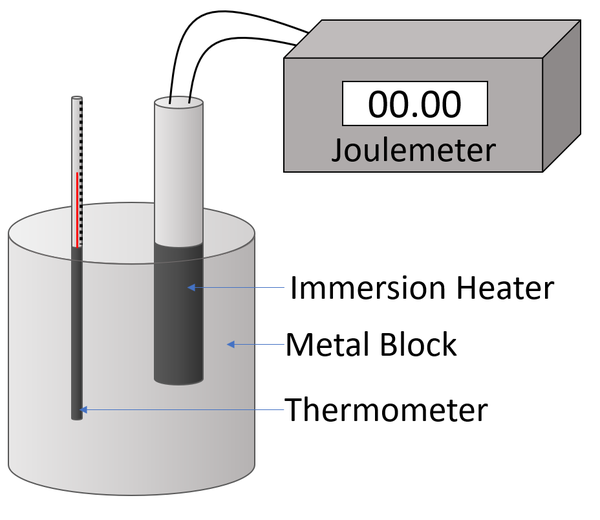
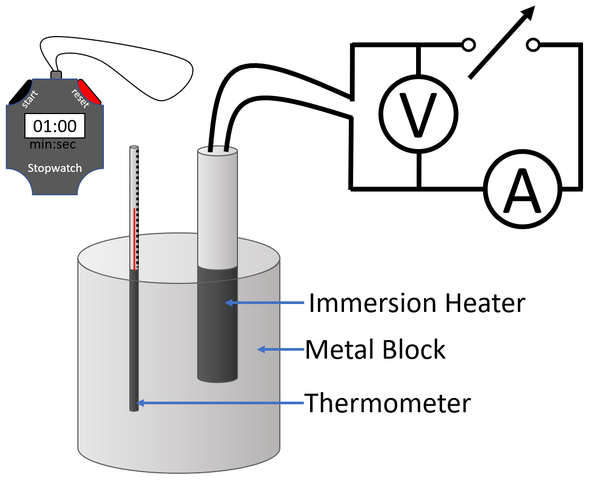
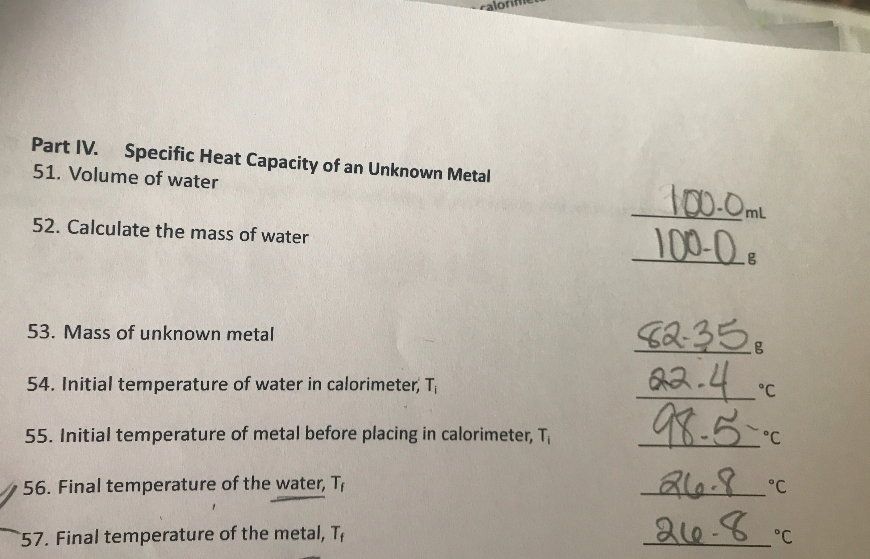LAB: Specific Heat of a Metal. Prelab question: MetalSpecific Heat (J/g ºC) Aluminum0.91 Iron0.46 Lead0.13 Silver0.23 Tin0.21 Titanium0.54 Zinc0.39 A. - ppt download
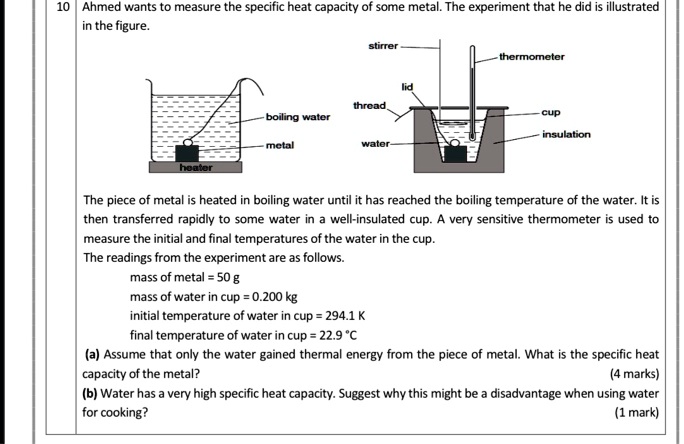
SOLVED: Ahmed wants measure the specific heat capacity of some metal. The experiment that he did is illustrated in the figure sunrer thermometer tnrcad boiling water insulation metal wator The piece of

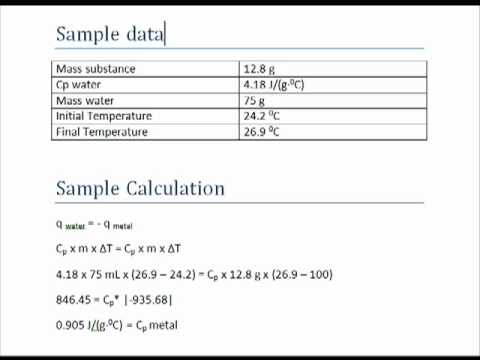
CALCULATING SPECIFIC HEAT CAPACITY Using lab data and your understanding of thermal energy! - ppt download

SOLVED:If 7.24 kJ of heat is applied to a 952 -g block of metal, the temperature increases by 10.7 . C . Calculate the specific heat capacity of the metal in J / g ' C.

A mass of 50 g of a certain metal at 150^0C is immersed in 100 g of water at 11^0C. The final temperature is 20^0C . Calculate the specific heat capacity of

LAB: Specific Heat of a Metal. Prelab question: MetalSpecific Heat (J/g ºC) Aluminum0.91 Iron0.46 Lead0.13 Silver0.23 Tin0.21 Titanium0.54 Zinc0.39 A. - ppt download