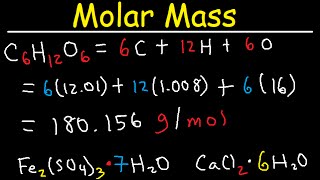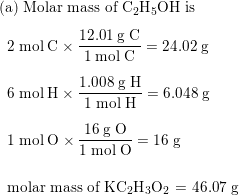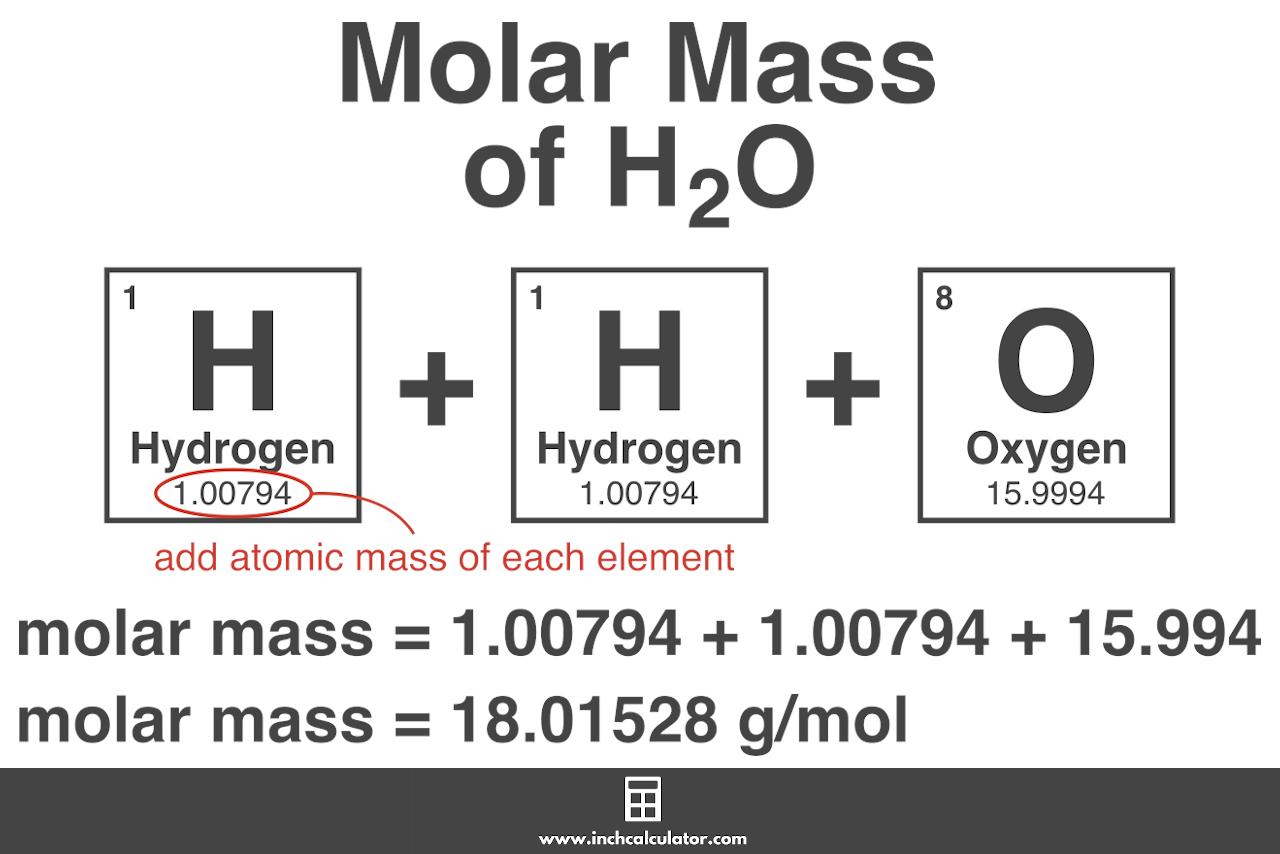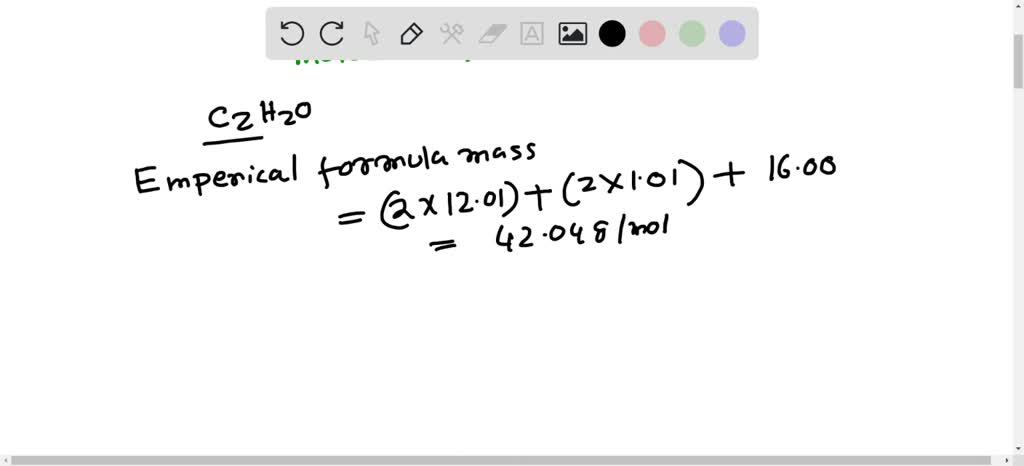
SOLVED: Calculate the molecular formula of a compound with the empirical formula C2H2O and a molar mass of 126.12 g/mol.

Calculate the mass of compound (molar mass = 256 g mol^-1 ) to be dissolved in 75g of benzene to lower its freezing point by 0.48 K (Kf = 5.12 K kg mol^-1 ) .